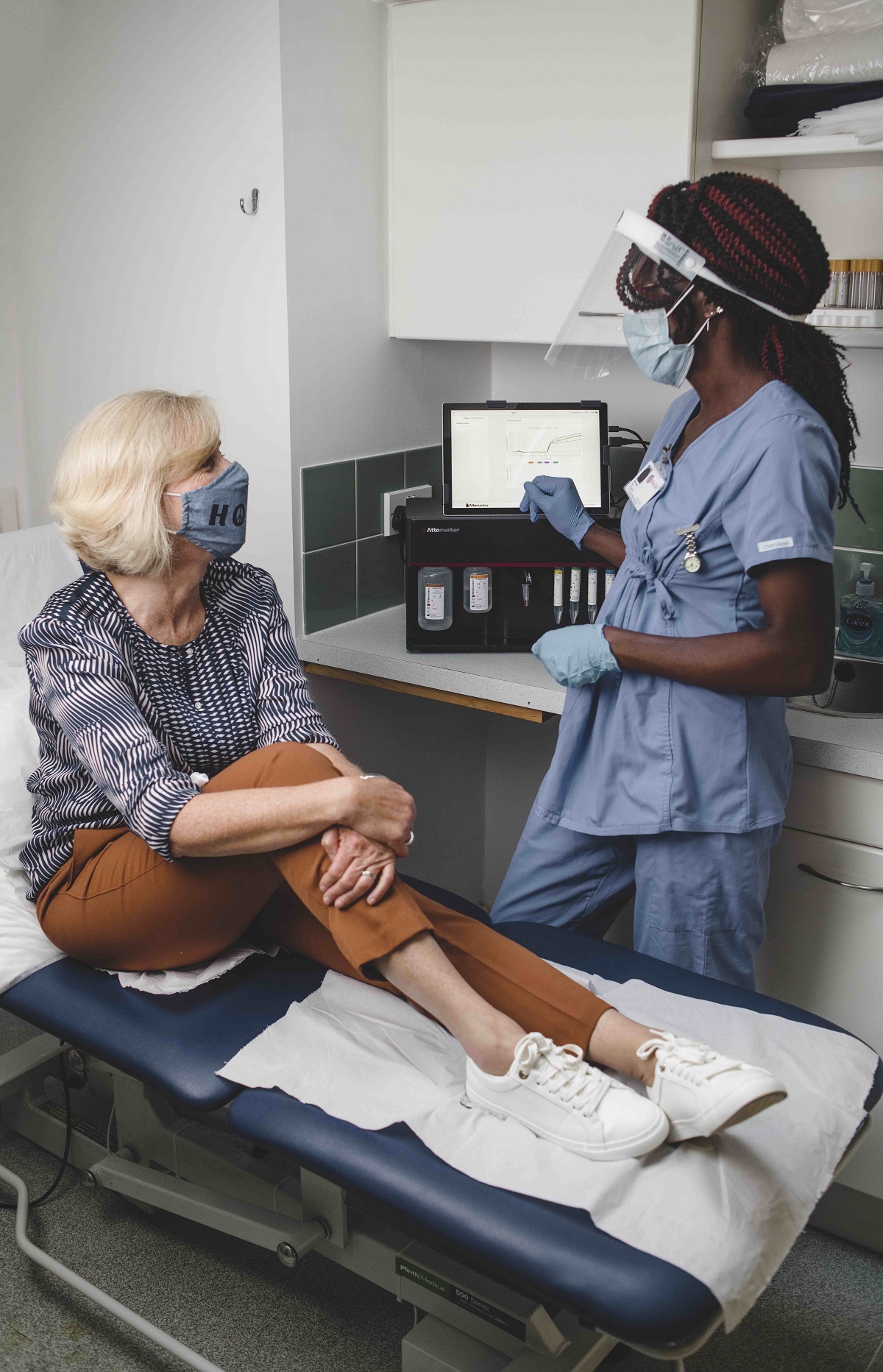
A British med-tech company founded at the University of Exeter has received an investment from Innova, the world’s largest COVID-19 test provider, to accelerate the rollout of its novel immunity test. Attomarker’s CE marked and MHRA accredited rapid COVID Antibody Immunity Test assesses an individual’s antibody levels and estimates when they will likely be on the ‘edge of immunity’; providing vital insight for patients and public health officials to better inform the timing of shots and other protective measures.
The device is the first of its kind to measure an individual’s immune response to a COVID-19 vaccination, booster, infection – or a combination of the immune triggers. With recent Covid cases in the UK at near-record levels, many may now have natural immunity to supplement the waning vaccine protection: some have good protection until the autumn, but others are more vulnerable.
Both companies believe widespread immunity testing would help public health bodies and governments distribute vaccines more efficiently whilst avoiding unnecessary shots for patients. The technology – currently available in selected clinics in the UK, Europe and the Caribbean – provides patients and clinicians with reliable, science-based information to make personalised healthcare decisions in just seven minutes.
The partnership will see Innova become an official distributor of the Attomarker product. Innova’s CEO hopes its commercial clout in the rapid test space will help accelerate Attomarker’s growth for immunity testing into other markets and overseas. Aside from COVID, the two companies soon hope to develop additional tests for other complex diseases, using the biomarker technology to address issues such as liver function, food allergies and fertility problems.
Professor Andrew Shaw, CEO and Founder of Attomarker, said: “Our test gives results in seven minutes and demonstrates an individual’s level of protection, whether they’ve had an infection in the last six months and when they’ll likely need their next shot.
“A ‘test to boost’ approach would deliver greater vaccine accuracy and could save millions whilst avoiding over-vaccination autoimmunity complications – where the body’s immune system attacks healthy cells.
“It’s about understanding what your immunity profile is – and how good your response has been – following a vaccine. There are some patients who have been poor responders to booster jabs and some who are ‘super responders’ – individuals who may not need a booster in 3-6 months and can wait longer than most of us.
“Armed with these data, people and governments can make informed choices about jabs and personal assessments around how they live out their daily lives, such as safe travel and return to work, leading to a more personalised and precise strategy for living with COVID. This strategic alliance with Innova accelerates this vision to a truly global level.”
CEO of Innova, Daniel Elliott, commented: “We are delighted to announce this partnership with Attomarker.
“Our team has been searching globally for advanced diagnostics technologies to manage the current pandemic, to make us better prepared for the next one and to improve public health worldwide. We were deeply impressed by the Attomarker platform technology and all it has to offer.
“We are excited about working with the Attomarker team to help them accelerate growth. Adding a much-needed immunity test into Innova’s COVID rapid test portfolio strengthens the pandemic management solution we offer to our global customers.”
The Medical Director of Innova, Dr Todd Malan, said: “I’m excited to work collaboratively with the scientists at Attomarker. Their technology has the potential to change healthcare diagnosis and treatment with a portfolio of near-patient rapid diagnostics that would lead to more effective clinical intervention.
“Most immediately, the unique Attomarker COVID-19 Antibody Immunity Test is fully quantitative, providing information on vaccine efficacy to variants and has highlighted the significance of immune defences in the nose for preventing infection. Attomarker’s technology has the potential to change healthcare diagnosis and treatment with a portfolio of near-patient rapid diagnostics that would lead to more effective clinical intervention.”
Prof Neil Gow, Deputy Vice-Chancellor for Research at the University of Exeter, said: “The novel and exciting Attomarker technology platform addresses a key gap in the diagnostic market and has the virtues of being rapid, precise, highly portable and adaptable. We are delighted to see the partnership with Innova develop to deliver tangible benefits for the community at large.”